Reports of research work funded by grants prior to 2012
Victoria University of Wellington
Enhancing human regulatory monocyte activation (2010/191)
AC La Flamme, D Chuluundorj, and S Harding
School of Biological Sciences, Malaghan Institute of Medical Research and Wellington Regional Hospital
Aims
This research project investigated the hypothesis that human monocytes can be activated into a “regulatory” phenotype with immune complexes or glatiramer acetate. To address this hypothesis, we analyzed in vitro responses of monocyte isolated from healthy subjects and determined if monocytes exposed to immune complexes or GA in a pro-inflammatory environment become regulatory monocytes.
Background
Recently, the role of regulatory monocytes in modulating immune responses has become evident. In particular agents that alter monocyte activation have been shown to be effective in immune-mediated disorders such as multiple sclerosis (MS). Glatiramer acetate, a common MS drug, is one such agent, but while it is clear that it alters monocyte responses, the regulatory phenotype has not been fully elucidated. Additionally, we have found that large immune complexes made from antibody-coated red blood cells are strong inducers of regulatory activation in murine macrophages. Thus we also wished to understand if this pathway promotes a similar activation state in human monocytes as glatiramer acetate.
Results
Our studies using monocytes from healthy subjects showed that both glatiramer acetate and immune complexes were effective at inducing a regulatory state; however, the phenotypes of these regulatory monocytes were distinct. In particular, we found that while both significantly reduced the expression of CD40, glatiramer.
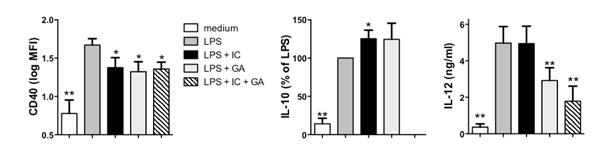
Figure 1: Regulatory monocytes induced by immune complexes (IC) or glatiramer acetate (GA) display distinct characteristics and a combination of both IC and GA produces an enhanced regulatory state. CD14+ monocytes were isolated from healthy subjects, primed overnight with IFN-γ, and stimulated for 24 hours with lippolysaccharide (LPS) in the presence or absence of IC, GA, or both. Shown are the means and SEM of CD40 (a; log mean fluorescence intensity), IL-10 (b; % of LPS alone), and IL-12 (c; ng/ml) from 21 subjects (n = 8 for IC + GA). * p < 0.05 compared to LPS alone and ** p < 0.01 compared to LPS alone by repeated measures ANOVA.
Acetate was more effective at reducing the production of the pro-inflammatory cytokine IL-12 whereas immune complex treatment significantly enhanced IL-10 production. Further analyses showed that cultures of purified CD14++CD16- or CD14+CD16+ subsets responded to the treatments similarly although the CD14+CD16+ and not the CD14++CD16- subset appeared to be responsible for the enhanced IL-10 production induced by the immune complexes. Finally, we found that the combined activation of monocytes with both immune complexes and glatiramer acetate showed significantly greater reductions in the pro-inflammatory mediators and markers as well as significantly increased regulatory mediators such as IL-10 suggesting that combining glatiramer acetate treatment with immune complex activation may improve the efficacy of this MS drug.
On-going research
Our on-going research investigates how these agents alter the responses of monocytes from MS patients. In particular, we are interested in understanding if monocytes from patients currently taking glatiramer acetate have different responses to monocytes from MS patients that do not take glatiramer acetate, and if regulatory activation can be improved by a combined treatment with glatiramer acetate and immune complexes. For this study we have recruited the help of Dr David Abernathy at Wellington Hospital.
Significance
The impact of this study, which investigates how best to activate monocytes to a regulatory state, is two-fold: 1) we aim to gain a greater understanding behind the immune factors involved in driving or suppressing MS and 2) we hope to identify new therapeutic strategies for treating MS and other immune mediated diseases.



