Reports of research work funded by grants prior to 2013
Institute of Environmental Science & Research Ltd
Is there a role for harman and norharman in tobacco dependence?
P Truman
Introduction
Harman (H) and norharman (NH), two naturally occurring monoamine oxidase inhibitors of tobacco smoke, are recognised as being of potential importance in tobacco dependence, and appear to have some significance in subtly affecting responses to nicotine (unpublished data). In the previous report (July 2011) we showed that half to a third of the MAO-A inhibition seen in tobacco was accounted for by H/NH.
Our remaining work had two objectives. The first was to examine whether H/NH concentrations built up in the brain to pharmacologically significant concentrations after longer term exposures, mimicking exposures during smoking. The second was to consolidate the initial findings on the proportion of MAO inhibitory activity that was due to harman and norharman, using a wider range of tobacco smoke condensate (TPM) samples.
Objective 1: Because an initial experiment indicated that H/NH concentrations in tissues were quite low, or were not being properly detected, we decided to do a more complex experiment than was first envisaged. We took advantage of behavioural experiments in which rats were exposed to nicotine alone, or nicotine plus H/NH at a similar concentration, relative to the nicotine, as is experienced by smokers. These self-administration experiments were conducted over a period of approximately 3 months. The rats were euthanased after the behavioural experiments were complete, and the brains dissected. The brain portions taken were: amygdyla, frontal cortex, hypothalamus, nucleus accumbens, striatum, substantia nigra and ventral tegmental area. Rats were sacrificed at different times after the last self- administration session, to see whether any recovery of MAO activity could be seen over time frames suggested by the literature. These brain samples were processed for determination of monoamine oxidase activity, and samples frozen at -80oC until their MAO activities could be individually determined. The results for rats sacrificed within 1 hour of their last self-administration session are summarised in Table 1. Results are expressed in pmoles of product produced per minute, per µL of sample. Each µL of sample contained 0.1 mg of homogenised brain tissue.
Table 1: Monoamine oxidase activity of brain tissue from rats chronically exposed to harman and norharman (H/NH) as part of a nicotine self-administration regime and sacrificed within 1/2 hour of completion of the self-administration experiments, n=3; rats which had self-administered nicotine only, n=2. (CoV = Coefficient of Variation).
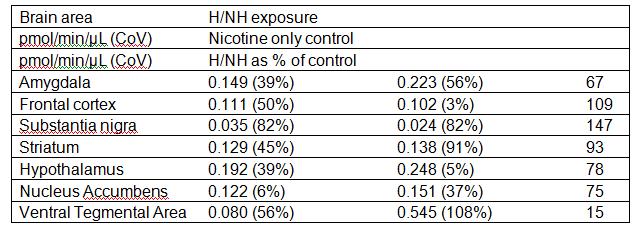
There was no significant change in MAO activity in any brain area, although the overall trend was towards lower MAO activity in brains of rats exposed to H/NH. Brain areas from a further 5 rats were also analysed. These were sacrificed at longer times after their last self-administration session (1 hour to 10 days). No pattern of increase in activity over time after the self-administration sessions could be discerned for any brain area (data not shown).
Brain and plasma samples were sent for instrumental analysis of H and NH content. The plasma samples were a time course of blood collection following a self-administration session, aiming to see whether we could find evidence for the half-life of H and NH in the blood, to confirm the literature values. The brain samples were pooled from two or three rats per brain region, from 5 brain regions: nucleus accumbens, hypothalamus, amygdyla, striatum, and ventral tegmental area.
Neither H nor NH was detected in any of the samples submitted. Use of a deuterated internal standard showed that the sample matrix was not suppressing ionisation to any significant effect, and that the detection limit was 10 ppb in these samples. This corresponds to the rats retaining less than 3 µg of H and NH (if evenly distributed through the body) at the time of sampling. Each rat dosed with harman and norharman, would have received 3 µg of harman and approximately 10 µg of norharman during these 2 hour self-administration sessions, and some rats were euthanased for brain collection within half an hour of their final self-administration session. If harman and norharman were able to accumulate in the brain, or had a long half-life in the body, they should have been detected.
The overall lessons learned from this experiment were that we would need to include fewer variables, and have a higher number of rats in each group in future experiments, but that also that we saw no convincing evidence for inhibition of MAO activity by H/NH following voluntary chronic exposure, as had been hypothesised.
Objective 2: We found, for six different TPM samples, that when harman and norharman are diluted to the extent that they only inhibit MAOA by 10%, TPM at the same harman/norharman concentration is inhibiting MAOA by over 50% (Table 2). Something other than harman/norharman alone is inhibiting the MAO enzyme.
Table 2: The % inhibition of MAO-A caused by TPM, and by equivalent harman/norharman (H/NH) mixtures. Each TPM is at 20 µM nicotine. Harman and norharman in TPM were analysed by liquid chromatography-mass spectrometry. All % inhibition figures are expressed relative to an “ethanol only” control.
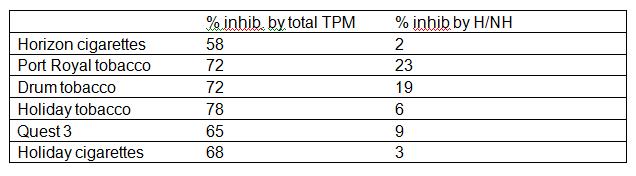
In these tobacco samples, the proportion of MAO inhibition due to harman/norharman varied from approximately 4% to over 30%. It is now clear that there is a sizeable proportion of the MAO inhibitory activity in tobacco smoke which is not due to harman and norharman.
In summary we found no evidence to support the hypothesis that harman and norharman are directly responsible for the observed decrease in MAO activity in the brains of smokers. The amounts of harman and norharman that heavy smokers are exposed to should, however, be sufficient to have some effects on brain monoamine oxidase activity.



