Reports of research work funded by grants prior to 2014
University of Otago Wellington
Evaluation of a panel of prognostic markers for colorectal cancer
R S Stubbs
Department of Pathology and Molecular Medicine
Summary of research: Colorectal cancer (CRC) continues to be a major cause of death in NZ despite improvements in management over the last 20 years. Discovery of a reliable prognostic biomarker(s) could have a major and positive impact on the management of and outcome from CRC. The Wakefield Biomedical Research Unit has been undertaking a biomarker discovery program in CRC and over the last six years has completed four biomarker discovery projects, the last of which was completed in August 2013. Each has involved proteomic comparisons of tissue derived from CRC patients with good and bad prognostic features using 2D-DIGE and gel free-mass spectrometry techniques of laser microdissected tissues (LMD). In the first study we examined the differences between the protein profile obtained from tissue at the invasive front of CRC, the main tumour body and liver metastases from the same patient. In the second, carried out by one of our PhD students, Hongjun Shi (PhD awarded August 2012), we identified cancer specific proteins from CRC (compared with normal colon tissue). In the third we identified proteins that are differentially expressed between stage II CRC patients who developed recurrence within five years and those who did not. In the fourth, we have identified proteins differentially expressed in tissue from rectal cancer in which there was or was not evidence of perineural invasion (PNI). PNI has been shown to be a very important prognostic feature in rectal cancer. Through this series of projects, employing a relatively novel approach, over 60 proteins have been identified as having potential value as prognostic markers. In the present study supported by the WMRF we sought to:-
Results: In our present study we have examined thirty potential biomarker protein candidates (Table 1) which were identified from our last two discover projects, mentioned above, in 409 CRC patients with known survival times noted at five year follow-up (Table 2).
We chose the 40 top priority candidates for which commercially available antibodies were readily available in 30. These 30 were examined in the present study.
Table 1: Proteins which were verified on 409 CRC tumour tissue samples
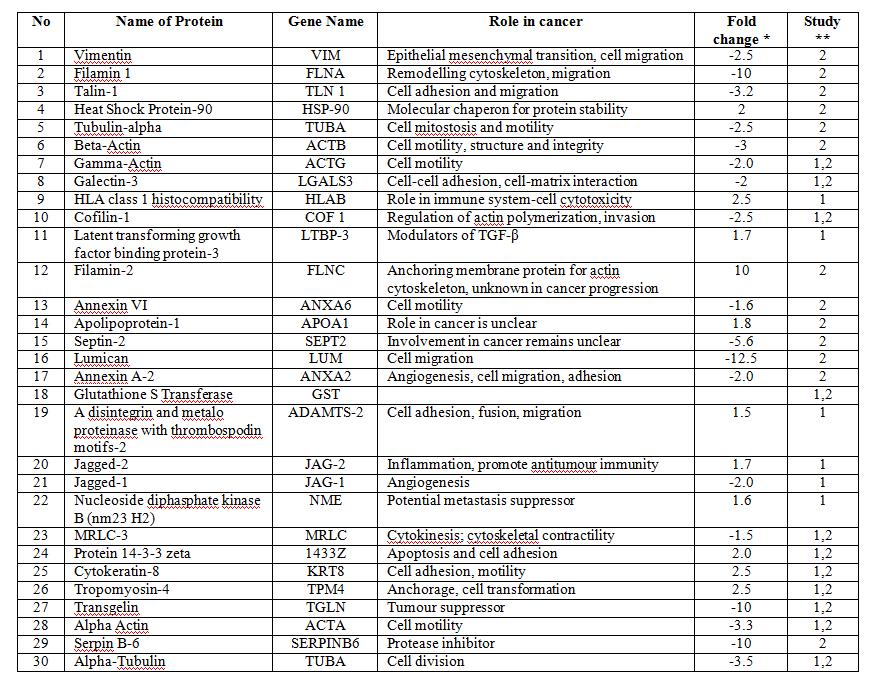 * Fold change is a ratio of less to more aggressive tumour i.e. non metastastatic tumour/metastatic tumour; PNI negative/PNI positive
* Fold change is a ratio of less to more aggressive tumour i.e. non metastastatic tumour/metastatic tumour; PNI negative/PNI positive
** Protein identified from 1) study of differentially expressed proteins between stage II CRC patients who developed recurrence within 5 years and those who did not and 2) study of differentially expressed proteins between rectal cancer tissue in which there was or was not evidence of PNI.
These candidates were examined in paraffin tissue microarrays from 409 patients with surgically removed CRC for whom survival times out to 5 years were known.
Table 2: Clinicopathological parameters profile of CRC samples from 409 patients
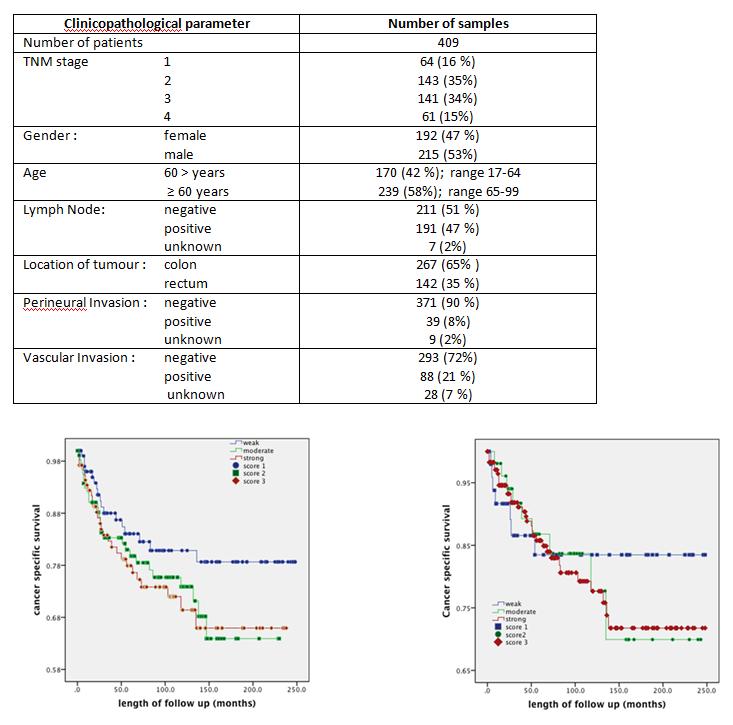 Fig 1. The survival curves shown here for two candidates (LTBP-3 (left) and Cytokeratin-18 (right) indicate that moderate or strong expression of each confers poor prognosis for patients.
Fig 1. The survival curves shown here for two candidates (LTBP-3 (left) and Cytokeratin-18 (right) indicate that moderate or strong expression of each confers poor prognosis for patients.
We include above in Fig 1 the survival curves of all 409 patients according to their expression (weak, moderate or strong) of two of our candidate proteins LTBP-3 and cytokeratin-18. Higher intensity of LTBP in tumour cells significantly correlated with histology of tumour (P= 0.0001); vascular invasion (P=0.0001); local recurrence (P= 0.021) and death from cancer (P=0.0001). Higher expression of Cytokeratin-18 significantly correlated with perineural invasion (P=0.044) and poor survival.
We are currently involved in a detailed statistical analysis of the clinical data and the expression of each of our candidate proteins to determine the potential clinical utility that any of our proteins might bring to the determination of risk of recurrent disease in patients with CRC. It is hoped that incorporation of one or more of our candidate proteins into routine pathological evaluation in the future will lead to improved selection of patients for adjuvant chemotherapy.
Acknowledgement: The Wakefield Biomedical Research Unit gratefully acknowledges support for our colorectal cancer research program received over a number of years from the Wellington Medical Research Foundation.



