Reports of research work funded by grants prior to 2015
Institute of Environmental Science & Research Ltd
How do meningococci defend against phagocytosis?
X Ren
In this study we set out to understand how some meningococcal strains can resist phagocytosis by macrophages. Meningococcal bacteria are the main causative agent for bacterial meningitis, which can cause inflammation of the meninges as well as septicaemia. This disease often causes long lasting disabilities and fatalities. Surprisingly, this bacteria can also be carried by healthy individuals without causing disease. Meningococci are highly diverse, only a small number of strains cause most of the diseases. We aim to understand how some strains cause disease and some do not by using a variety of cellular and molecular techniques. We found recently, using differentiated THP-1 cells as a model system for macrophages, that some carriage-associated isolates were phagocytosed at a higher rate than some disease isolates. Thanks to WMRF’s Grant in Aid of Research programme we were able to extend these studies.
1. Investigate in more detail the interactions between meningococcal isolates and phagocytes by live/dead and inside/outside staining techniques
In this objective we aimed to use cell biological techniques to dissect the timing of phagocytosis better as well as to distinguish live vs. dead bacteria inside of differentiated THP-1 macrophages. Unfortunately, we were not able to get the live-dead staining to work in our assays. We found that sytox-green and sytox orange stained meningococci even when the meningococci are intact. We tried a range of concentrations, including that which was used for studying similar phenomenon in Neisseria gonorrhoeae, but we could not distinguish heat-killed bacteria from live bacteria. One reason for this could be is that we have to fix the bacteria before examining on a microscope due to the pathogenicity of meningococci. In the Neisseria gonorrhoeae study, bacteria samples were observed without fixing. We have tried washing the samples extensively before fixation, however we still had all bacteria being slightly stained making the calling of live vs dead difficult. We did use plating technique to investigate killing by macrophages. These results are preliminary, we found that we can recover more bacteria from macrophages that were infected by disease-associated strains (after gentamicin treatment to kill all external bacteria) suggesting that small numbers of disease-associated strains were phagocytosed by these macrophages but were able to survive and multiply.
We were able use the inside-out staining technique to confirm what was found using plating technique (Fig. 1).
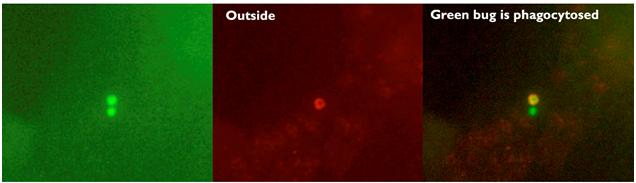
Fig. 1. Example of inside-out staining technique
2. Gain insights into the nature of the bacterial factor by mutant analysis
We used mutagenesis approach to discover factors that may be important for resisting phagocytosis (Fig. 2). Available literature as well as our own genomic and transcriptomic data was utilised to guide us in this process. We used the mutants that we generated in previous studies, as well as new mutants generated for this study. We found that similar to previous studies, the capsule (98-254 P10 and 98-254delsiaDb) has a minor role in preventing phagocytosis. Unlike in other bacteria, deleting genes encoding lipooligosaccharide modifying enzyme lgtA and major porin protein porB did not enable engulfment by macrophages. Lactoferrin-binding protein, lbpAB, and pili structural protein pilS also had a slight effect on phagocytosis. We have recently found that TNF-alpha level in the culture media of differentiated THP-1 cells that were infected with isolates which can be phagocytosed, were much higher than with isolates that cannot be effectively phagocytosed. In the future we could use this method to screen mutants more quickly. This part of the study shows that resisting macrophage phagocytosis is likely to be multigenic.
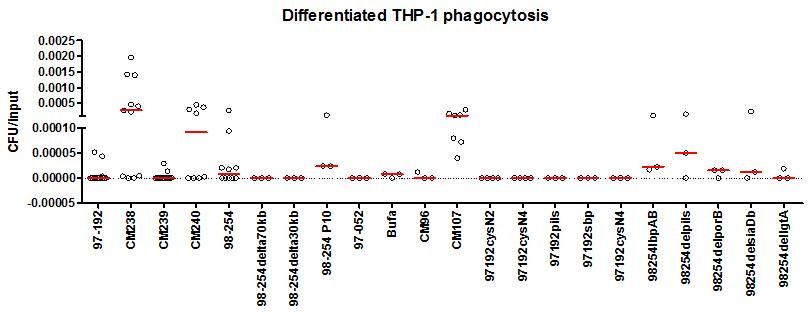
Fig. 2: Rate of phagocytosis in disease strains and carriage (indicated by CM) and mutants.
In conclusion we have used cellular techniques to confirm the results found using plating and further that some extracellular structures, such as pili and capsule, are more important in preventing phagocytosis than other extracellular proteins. Unfortunately we were not able to use live-dead staining to examine phagocytosis further. We used plating technique and found that disease-associated isolates may be able to survive phagocytosis by macrophages. We have also found measuring TNF-alpha levels in infected macrophages maybe a more efficient way to screen for mutants that allows disease-associated isolates to be phagocytosed.



