Reports of research work funded by grants prior to 2016
Institute of Environmental Science & Research Ltd
Nutritional Intervention Strategies for Allergic Skin Inflammation
LWJ van den Elsen
Background
This project was devoted to identifying pathways involved in the prevention and/or suppression of allergic skin inflammation by a synbiotic diet containing probiotics (beneficial bacteria) and prebiotic fibres (supporting the growth of beneficial bacteria). We utilised a mouse model of atopic dermatitis (AD) induced by topical application of the vitamin D analogue MC903 on the ears. The outline was based on preliminary results that had shown that the synbiotic diet is effective in the suppression of allergic skin inflammation. Hence, this diet seemed a potential therapeutic approach to prevent or treat AD and the subsequent atopic march. Both have an enormous impact on the quality of life of patients and are a great financial burden for the New Zealand society.
Results
As explained in our previous report, the results obtained with different batches of the synbiotic diet in the AD model have been inconsistent. Therefore we tried a different way of delivering probiotics daily in a bolus preparation to ensure administration of live bacteria. Unfortunately this approach did not result in suppression of AD symptoms as measured as the allergic skin response (Fig 1).
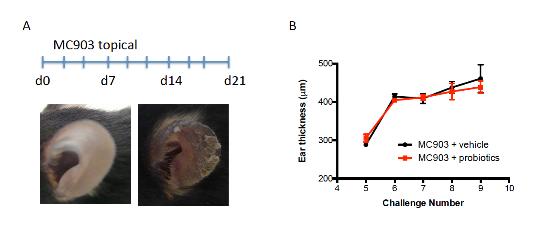
Figure 1 Administration of a probiotic mixture does not improve the allergic skin response in a mouse model for AD. (A) Mouse model of MC903-induced AD with phenotype of ear skin at d0 and d21 of treatment (B) Allergic skin response depicted as ear thickness for both vehicle and probiotic treated mice during MC903 treatment.
We have also examined the potential efficacy of probiotic administration in another mouse model of allergic disease. The MC903 model is a very strong inducer of AD and the induction involves multiple pathways and cell types. Besides allergic skin inflammation, allergic asthma contributes to the atopic march as well. Therefore, the probiotic mixture was tested in a mouse model for house dust mite-induced allergic airway inflammation (Figure 2A). Probiotics were not effective in reducing the concentration of IgE in the serum (Figure 2B) or in preventing allergic airway inflammation as studied by flow cytometery in the bronchoalveolar lavage fluid (BALF) (Fig 2C, D). Other BALF cell subsets including dendritic cells, neutrophils, T and B cells did not differ between groups either (data not shown)
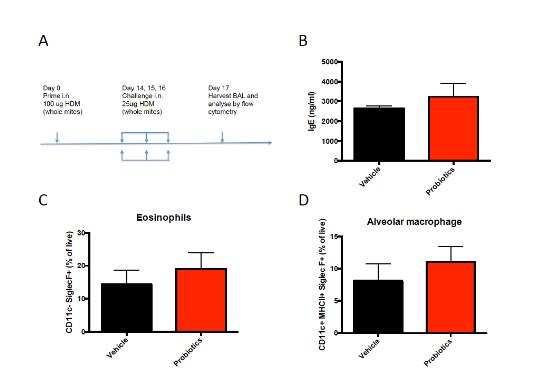
Figure 2 Administration of a probiotic mixture does not affect allergic airway inflammation. (A) Mouse model for house dust mite-induced allergic airway inflammation. (B) Concentration of total IgE in serum obtained one day after the last intranasal challenge. (C) Percentage of eosinophils and (D) alveolar macrophages in BALF of HDM sensitized and challenged mice. BALF = bronchoalveolar lavage fluid; HDM = house dust mite.
Conclusion
The synbiotic diet was not effective in suppressing allergic skin inflammation in a mouse model for AD. Daily bolus administration of probiotics also did not prevent and/or reduce symptoms in this model, nor a model for house dust mite-induced allergic airway inflammation. Therefore we conclude that the specific synbiotic diet and probiotic mixture examined are not a promising approach to prevent or treat AD and the atopic march. In the future we will focus on other nutritional intervention strategies to improve allergic outcome.



